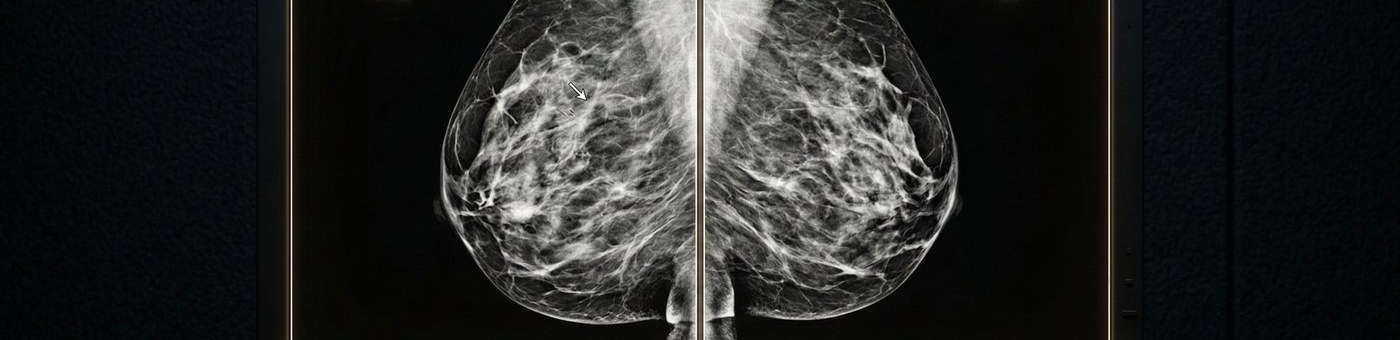
Mammography Reading Services
AI-Assisted BI-RADS Reporting
What Sets Our Mammography Program Apart
Every aspect of our mammography teleradiology service is built around federal compliance requirements, clinical accuracy, and the specific needs of breast imaging programs.
MQSA-Compliant Radiologists
All mammography reads are performed by radiologists who meet or exceed MQSA (Mammography Quality Standards Act) physician requirements — including minimum annual read volumes and continuing medical education in mammography. Compliance documentation is available on request.
2D and 3D Tomosynthesis Reads
We interpret both conventional 2D digital mammography and 3D digital breast tomosynthesis (DBT) — including combo mode studies (2D + 3D acquired together). Our readers are experienced on GE, Hologic, Siemens, and Fujifilm mammography systems.
Screening and Diagnostic Coverage
Screening mammography reads, diagnostic mammography workups, and additional views (spot compression, magnification, rolled views) are all covered. Diagnostic reads include clinical correlation with patient history provided by your center.
ACR BI-RADS Structured Reporting
Every mammography report uses ACR BI-RADS 5th Edition structured reporting — including BI-RADS assessment category, composition category, findings description, and recommendation. Reports are formatted to your PACS or RIS requirements.
Mammography Reading, by the Numbers
Key metrics across breast imaging center partners
AI
Assisted detection on every study
< 4 hr
Diagnostic mammography turnaround
100%
MQSA-compliant reading physicians
ACR
BI-RADS 5 structured reports
MQSA-Compliant Mammography Reads
Understanding MQSA Requirements for Teleradiology Mammography
The Mammography Quality Standards Act (MQSA) establishes specific requirements for radiologists who interpret mammography studies — requirements that go beyond standard board certification. Any teleradiology provider offering mammography reads must meet these federal standards, and your facility remains responsible for ensuring the interpreting physicians comply.
At Natoe AI, all mammography reads are performed by physicians who meet MQSA's initial qualification and continuing experience requirements. This includes minimum annual interpretive volumes (960 mammographic examinations per 24 months) and ongoing mammography-specific CME. We maintain MQSA compliance documentation for each reading physician and provide it to client facilities on request.
MQSA also requires that mammography reports be issued directly in the patient's name and that results be communicated to the patient within 30 days. Our reporting workflow supports both requirements — reports are structured per ACR BI-RADS guidelines and formatted for direct patient communication when needed.
Frequently Asked Questions
Are your mammography readers MQSA-compliant?
Yes. All radiologists reading mammography for Natoe AI meet MQSA physician qualifications — initial training requirements, annual interpretive volume minimums, and ongoing mammography CME. Compliance documentation is available to client facilities on request.
Do you read 3D tomosynthesis (DBT) studies?
Yes. We read 2D digital mammography, 3D digital breast tomosynthesis, and combo-mode studies acquired on Hologic, GE, Siemens, and Fujifilm systems. Our readers have routine clinical experience with DBT interpretation.
What is your turnaround time for mammography reads?
Screening mammography is typically reported within 24 hours of study receipt. Diagnostic mammography workups are prioritized and typically reported within 4 hours. Same-day turnaround is available for diagnostic studies when clinically indicated.
Do you provide both screening and diagnostic mammography reads?
Yes. We cover screening mammography, diagnostic mammography, and additional views (spot compression, magnification, rolled views). Diagnostic reads include correlation with the clinical history provided by your facility.
What reporting format do you use for mammography?
All mammography reports use ACR BI-RADS 5th Edition structured reporting, including BI-RADS assessment category (0–6), breast composition (A–D), finding descriptions, and recommendations. Reports can be formatted to integrate with your RIS system.
Are you hiring breast imaging radiologists?
Yes. Natoe AI hires MQSA-certified, US-licensed breast imaging radiologists for remote reading. We post our active openings on our careers page — see the Remote Breast Imaging Radiologist role for current details, or browse all open radiologist roles.